TRELAB EXE. E: Write balanced equations for the dichromate and hypochlorite oxidations of cyclohexanol to cyclohexanone, and for the per- manganate oxidation of cyclohexanone to adipic acid.
Q: LiOH + KCl → LiCl + KOH I began this reaction with 20 grams of lithium hydroxide. What is my...
A:
Q: topic: matter do you think that each sample occupies space? write the reason(s) for each sample sm...
A: Yes, since the two items listed above is an example is matter which occupies space while the light a...
Q: Properly (i.e., systematically) name the following 1. PAS3
A: The name of a compound can be done by the IUPAC rule. According to which, in an inorganic compound, ...
Q: Which of the following reactions has a negative entropy change? Group of answer choices NH4Cl(s) → N...
A:
Q: Rank the gasses from highest to lowest global warming potential. (1 being the highest, 4 being the l...
A: Due to global warming, the temperature of our earth increases day by day.
Q: B: Identify water as either a Bronsted-Lowry acid or a Bronsted-Lowry base of the reactions shown be...
A: The reactions given are,
Q: The initiation step of radical halogenation of alkanes involves homolytic bond formation. Select the...
A: Radical halogenation is the substitution of hydrogen atom of alkane by a halogen atom.
Q: A mixture was found to contain 1.50g NH4CI, 0.80 g NaCI, and 1.20 g SiO2. What is the percentage of ...
A: Given: Mass of NH4Cl = 1.50 g Mass of NaCl = 0.80 g Mass of SiO2 = 1.20 g
Q: Balance the following combustion reaction: (Write whole-number coefficients in each box.) 2,3,4-trim...
A: The Chemical reaction follows the law of conservation of mass. According to the law of conservation ...
Q: Cyclohexane derivatives exist primarily in the most stable of the available chair conformations. Giv...
A: Most stable chair conformation is that faces least strain interaction. Thus to draw the most stable...
Q: Balance the following equations: a) Fe2O3 + HNO3 = Fe(NO3)3 + H2O b) NO2 + H2 = ...
A: A chemical equation should always be balanced. That is, the number of atoms on both the sides of the...
Q: The compound known as diethyl ether, commonly referred to as ether, contains carbon, hydrogen, and o...
A: a) Given: mass of sample used = 2.751 g. Mass of CO2 produced = 6.533 g. Mass of H2O produced = 3.34...
Q: 2. Complete the table below. Indicate what is observed if each test is positive. State what this res...
A: Completion of the table in which the indication what is observed if each test is positive is given b...
Q: Iridium (Ir) has a face-centered cubic unit cell with an edge length of 383.3 pm. Calculate the dens...
A: Given: Edge length of the unit cell = 383.3 pm = 383.3 × 10-10 cm. (Since 1 pm...
Q: The Latin word parum affinis means fatty. Select the correct response: O True False
A: Given statement is The Latin word parum affinis means fatty.
Q: Activity 1: S OLVE ME BABY ONE MORE TIME! Directions: Calculate the change in energy for the followi...
A:
Q: If 87 J of heat are added to a balloon, causing it to expand from 1.0 L to 1.4 L under 0.97 atm of p...
A: Given: Required heat = 87 J Initial volume = 1.0 L Final volume = 1.4 L External pressure = 0.97 at...
Q: mem 101 G periodic table - Google Search How many moles of lithium hyd x Question 6 of 8 How many mo...
A: given moles of O2 = 0.78 mol
Q: Combustion of alkanes is an oxidation process. Select the correct response: O True False
A: Yes, Combustion is an example of an oxidation reaction. Many fuels or hydrocarbon contain carbon and...
Q: Rewrite this measurement with a simpler unit, if possible. kg-m 4.3 3. m
A: Given measurement is : Rewrite this measurement with a simpler unit, if possible = ?
Q: (2) Write the preliminary one pair of electron dots between each pair of bonded atomic cores. Figure...
A: Valence electrons present in each H atom = 1 Number of H at...
Q: FeBr2 + 2 KCl → FeCl2 + 2 KBr What is my theoretical yield of iron (II) chloride if...
A:
Q: How many moles of solute are present in 1.5 L of 0.20 Molarity Na2SO4 ?the weight of solute is 43 gr...
A: Volume=1.5L Molarity=0.20M Weight of solute=43grams
Q: 2. Expand the following bond-line representati ons to show all the atoms inclu ding C and H, (a) (6)...
A:
Q: Fill in the gaps in the following table: Symbol P3- Protons 35 49 Neutrons 45 66 118 Electrons 46 76...
A:
Q: Identify each of the following groups as an activator or deactivator and as an o,p-director or m-dir...
A: Given : We have to explain whether the gives groups are activator or deactivator.
Q: Answer the following questions about preparing the same buffer solution using three different method...
A:
Q: and cis trans
A: The 1,3 diaxial interactions are the only factor that destabilizes the compound. We have to answer ...
Q: 3. Predict products stereochemistry and mechanisms of the following reactions. CO2H A = .CO2R Ph N' ...
A: The given substrate is an enolizable aldehyde which in the presence of the chiral catalyst (R-prolin...
Q: The following set of data was obtained by the method of initial rates for the reaction: S2O82-(aq)...
A:
Q: The gas phase decomposition of nitrosyl chloride at 400 K NOCI(g)- →NO(g) + ½ Cl2(g) is second order...
A:
Q: Freezing Point Depression: Molar Mass of a So Report Sheet Unknown solid number 942g 0.1949 2.94°C ....
A: A numerical problem based on mole concept that is to be accomplished.
Q: Predict the major product of this reaction. K2Cr207 H3O+ II HO2C. CO2H HO `OH II IV HO2C. HO
A: option D is correct
Q: The structure given is Escitalopram, an active enantiomer of citalopram and works as an anti-depress...
A:
Q: n the calorimetry experiment was used to measure the heat of reaction for an endothermic process, wh...
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Explain how you could separate an equimolar mixture of Compound X, Y and Z. w chart). он Compound X ...
A: This problem needs a practical knowledge. It is a practical thing to separate them.
Q: MİCRONS 5.5 2.5 100 26 27 28 2,9 3 3.5 4,5 11 12 13 90 70 50 40 30 20 1773 cm 10 3800 300 3400 3200 ...
A:
Q: Correct the ones that are marked wrong:
A: 1 (b) BaSO4(s) + HCl (aq) -----------> No rea...
Q: Consider this two-step mechanism for a reaction. NO2 (g) + Cl2 (9) →CINO2 (g) + Cl (g) Slow NO2 (9) ...
A: We know a faster reaction have lowest activation energy and a slower reaction should have highest ac...
Q: Phosphorus pentachloride decomposes to phosphorus trichloride and chlorine gas at elevated temperatu...
A:
Q: Calculate the new boiling point of a solution containing 5.27 g of sucrose (C12H22011) and 45g of wa...
A: Molality is defined as number of moles of solute dissolved per kg of solvent.
Q: What is ΔS for the boiling of C2H5OH? Use the data given below. Assume that enthalpy and entropy val...
A: A molecule or an atom is in constant motion at normal temperature, the motion also includes vibratio...
Q: The following reaction is monitored as a function of time: AB→A + B. A plot of 1/[AB] versus time yi...
A: Since you have posted questions with multiple sub-parts, we are entitled to answer the first 3 only....
Q: 1. In classical Physics and general Chemistry, matter is any substance that has mass and takes up sp...
A:
Q: Calculate [OH¯] for a solution where [H,O*] = 0.00379 M. %3D [OH¯] = M
A: For an aqueous solution, we know that the production of the concentration of the hydrogen ions and t...
Q: CI
A:
Q: If the K of a monoprotic weak acid is 1.8 x 10-6, what is the pH of a 0.35 M solution of this acid?
A: Given: Ka of weak monoprotic acid (Assuming HA) = 1.8 × 10-6 And the concentration of HA = 0.35 M
Q: Rank the compounds in each group according to their reactivity toward electrophilic substituti 1 = m...
A: Rate of Electrophilic substitution reaction increases if benzene ring is attached to the electron wi...
Q: Conformers like geometric isomers are stable and can be isolated. Select the correct response: True ...
A:
Q: Be + 2 HCl --> BeCl2 + H2 My theoretical yield of beryllium chloride was 10.7 grams. I...
A: Since you have posted multiple questions as per guidelines we can answer only one per session . If y...

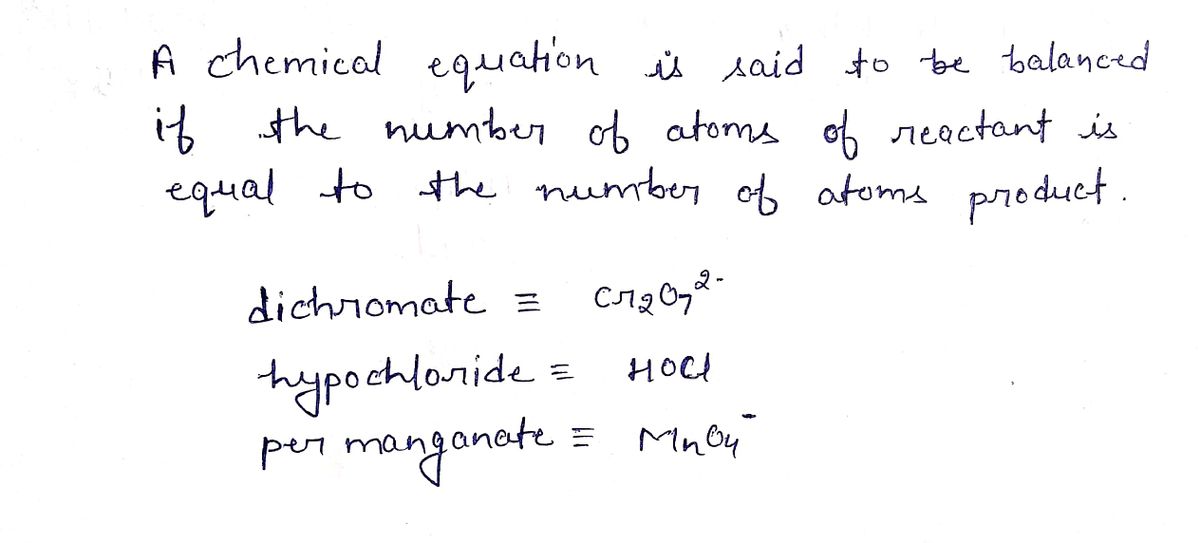
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- When a compound like napihthalene C1oHa is dissolved in t-butyl methyl ether, then that solution is extracted with 3 M NaOH, and then the resulting basic aqueous layer is acidified with 6M HCI, what happens to the acidified aqueous layer? a) The naphthalene stays in the the aqueous layer as C10Ha O b) The naphthalene precipitates out as a solid, C10He O C) Nothing happens to the aqueous layer other than a dramatic raising of the pH d) The naphthalene stays in the aqueous layer as C10H>Na O e) Nothing happens to the aqueous layer other than a dramatic lowering of the pH O) The acid precipitates out as a solid, C10H>NaChrysanthemic acid occurs as a mixture of esters in flowers of the chrysanthemum (pyrethrum) family. Reduction of chrysanthemic acid to its alcohol followed by conversion of the alcohol to its tosylate gives chrysanthemyl tosylate. Solvolysis alcohols. of the tosylate gives a mixture of artemesia and yomogi HO НО Chrysanthemic acid Chrysanthemyl alcohol OH TsO DMSO Chrysanthemyl tosylate Artemesia alcohol Yomogi alcohol Propose a mechanism for the formation of these alcohols from chrysanthemyl tosylate.could you synthesize isopropyl propyl ether, using isopropyl alcohol as the only carbon-containing reagent?
- preparation from 1-butanol to butylamineWrite complete reaction (with structures) for the following reactions: (a) Esterification of ethanoic acid with propanol (b) Reduction of 4-methyl-2-pentanone0.2 gm of ethanol was acetylated with acetic anhydride in presence of pyridine (1:3), neutralization of the resulted solution required 20 ml of 0.1 N NaOH, while the blank titration reqiuered 40ml of NaOH. Calculate the percentage of hydroxyl group in this sample ?
- Show how to convert carboxylic acids to other functional groups, and devisemultistep syntheses using carboxylic acids as starting materials and intermediates.Explain how acid chlorides are used as activated derivatives of carboxylic acidsGive TWO reagents to distinguish cyclohexanone from hexanal.propanal propanoic acid N-ethylpropanamide -HO- AA acidified KMN0, H;C NH2 NH CH3 H3C H3C BB H3C- DD HJ Ni он H;C propan-1-ol CC (ii) Name the type of chemical reaction for the formation of compound CC. Namakan jenis tindak balas kimia bagi penghasilan sebatian CC.

