
Concept explainers
Which compound in each pair is the stronger acid?
a.
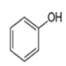 or
or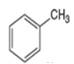
Trending nowThis is a popular solution!

Chapter 2 Solutions
Organic Chemistry (6th Edition)
- Which base, CH3NH2 or (CH3)2NH, is the stronger base? Which conjugate acid, (CH3)2NH2+ or (CH3)2NH3+, is the stronger acid?arrow_forwardWhat is the relative trend in acidity and pKa of the two compounds? a. Structure I is the most acidic, and Structure I has the highest pKa. b. Structure I is the most acidic, and Structure I has the lowest pKa. c. Structure II is the most acidic, and Structure I has the highest pKa. d. Structure II is the most acidic, and Structure I has the lowest pKa.arrow_forwardIn each equilibrium, label the stronger acid, the stronger base, the weaker acid, and the weaker base. Also estimate the position of each equilibrium. (a) CH3CH2O + CH3CCH CH3CH2OH + CH3CC (b) CH3CH2O + HCl CH3CH2OH + Cl (c) CH3COOH + CH3CH2O CH3COO + CH3CH2OHarrow_forward
- Weak Base NH (CH) N (CH3)2NH H₂NNH₂ Which one of the following is the strongest weak base? A NH3 Kb B 1.8 x 10-5 (CH3) N 6.4 x 10-5 5.4 x 10-4 C (CH3)2NH 1.3 x 10-6 D H₂NNH₂arrow_forwardWhich is the strongest acid? OA. В. FO | CH;CHCH,ČOH CH;CH,CHCOH CH3CH,CHČOH CHCHC-COH F Farrow_forwardWhich is the stronger acid? a. CH3CH3 or HC CH b. H2C CH2 or HC CH c. H2C CH2 or CH3CH3arrow_forward
- A ΝΗ most basic B > V с NH₂ V NH₂ H3CO E least basic NH₂arrow_forwardWhich is a stronger base? a. HS− or HO− b. CH3O− or CH3NH c. CH3OH or CH3O d. Cl− or Br− e. CH3COO− or CF3COO− f. CH3CHClCOO− or CH3CHBrCOO−arrow_forward4. Rank the following compounds from strongest acid to weakest acid: ОН ОН TOH JOH I Y A B C Darrow_forward
- What is the major product of this reaction? Ph. HO H,C H Ph H3C Ph Ph H3C Ph Ph Ph H H3C Ph Ph H2C H Pharrow_forwardWhich is the stronger base? a. Br- or I- b. CH3O- or CH3S-c. CH3CH2O− or CH3COO−d. H2C =CH e. FCH2CH2COO− or BrCH2CH2COO− f. ClCH2CH2O− or Cl2CHCH2O−arrow_forwardWhich of the following compounds is a strongest base? NH₂ O₂N 01 Oll O III ON OV NH₂ = II E III NH₂ IV NH₂ NH₂arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


