
Interpretation:
The
Concept Introduction:
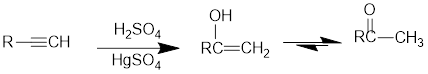
Acid Catalysed addition of water: When water is added to alkyne in the presence of an acid, the product formed will be an enol. Enol contains a double bond and a
If a carbonyl group is bonded to two alkyl groups, it is called as a ketone. The enol formed in the acid catalysed addition of water will be easily converted into a ketone.
Conversion of terminal
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Essential Organic Chemistry (3rd Edition)
- On synthesis of esters via nucleophilic acyl substitution: Write the chemical equation involved in the reaction between the excess acid and NaHCO3. Given this, briefly explain why NaHCO3 is preferred over NaOH for the neutralization of excess acid. Also, explain how excess alcohol was eliminated from the crude product.arrow_forwardDefine Enamine Formation from an Aldehyde or a Ketone ?arrow_forwardPredict the products formed when cyclohexanone reacts with the following reagents.ethylene glycol and p-toluenesulfonic acidarrow_forward
- Draw the keto and enol forms of (a) propanal and (b) 3-pentanone.arrow_forwardWhy can’t 2-methyl-2-propanol be prepared by the reduction of a carbonyl compound?arrow_forwardHydration of aldehydes and ketones can be catalyzed by acid or base. Bases catalyze hydration by: protonating the carbonyl oxygen making the carbonyl group more electrophilic employing hydroxide ion, which is a better nucleophile than water making the carbonyl group less electrophilic shifting the equilibrium position of the reaction to favor productsarrow_forward
- Give the products formed when benzaldehyde and benzoic acid are treated with the given reagents. 1 mole CH3OH, H+ LiAlH4 then H2O, H+arrow_forwardThe substance provided can be prepared by a nucleophilic addition reaction between an aldehydeor ketone and nucleophile. Identify the reactants from which it was prepared. If the substance is an acetal, identify the carbonyl compound and the alcohol; if it is an imine or enamine, identify the carbonyl compound and the amine.arrow_forwardIn which condition can carboxylic acids undergo nucleophilic acyl substitution reactions?arrow_forward
- What is the major advantage of base-catalyzed hydrolysis of esters over acid-catalyzed hydrolysis?arrow_forwardWrite chemical reaction of the epoxide preparation by reacting cis-2-pentene with a peroxycarboxylic acid. peroxycarboxylic acidarrow_forwardWhat is the reaction mechanism for formaldehyde and phenol under acidic conditions.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

