
Concept explainers
(a)
Interpretation:
The molecular orbital diagram of
(a)
Explanation of Solution
The electronic configuration is,
The molecular orbital diagram of
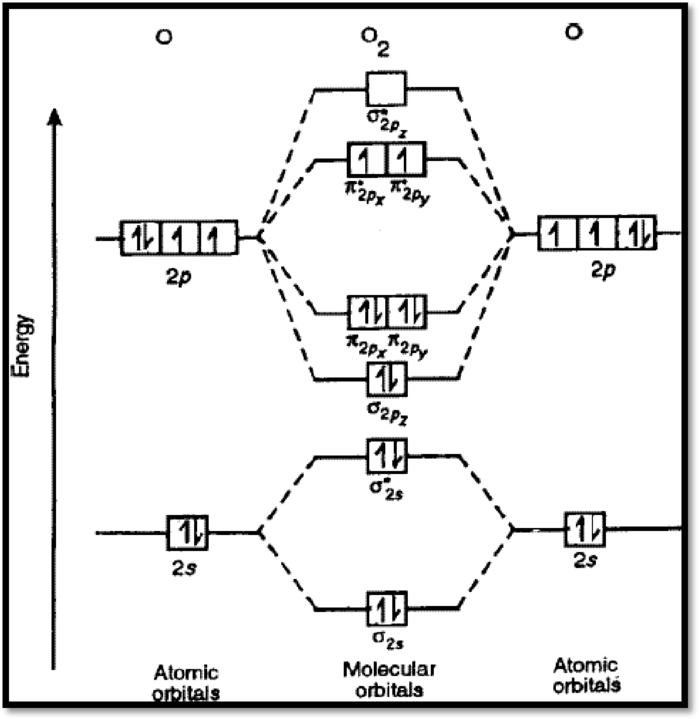
Figure 1
The bond order of
The bond order of
The magnetic nature of
(b)
Interpretation:
The molecular property of oxygen that is explained by its molecular orbital diagram but not by Lewis structure has to be given.
(b)
Explanation of Solution
The molecular property of oxygen that is explained by its molecular orbital diagram is magnetic property.
The Lewis structure of oxygen is,

From the Lewis structure, it can be seen that all electrons are paired, and it is diamagnetic in nature. However, the molecular orbital diagram of oxygen (figure 1) shows the presence of two unpaired electrons and therefore, oxygen is paramagnetic.
(c)
Interpretation:
The nature of highest occupied molecular orbital in oxygen has to be given.
(c)
Explanation of Solution
The electronic configuration of oxygen in the ground state is
(d)
Interpretation:
The bond order and magnetic property in peroxide ion and superoxide ion has to be given.
(d)
Explanation of Solution
The formula of peroxide ion is
The formula of superoxide ion is
Superoxide ion
The molecular orbital diagram of
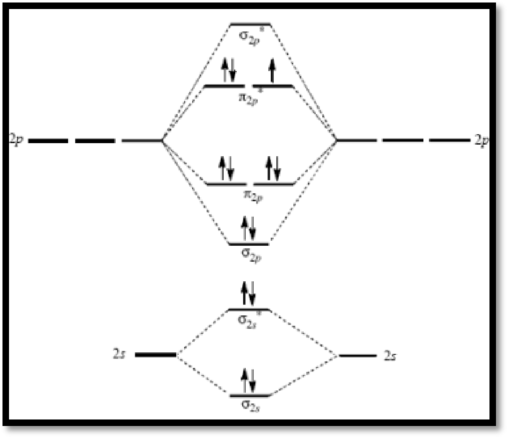
Figure 2
The bond order of
Superoxide ion
Peroxide ion
The molecular orbital diagram of
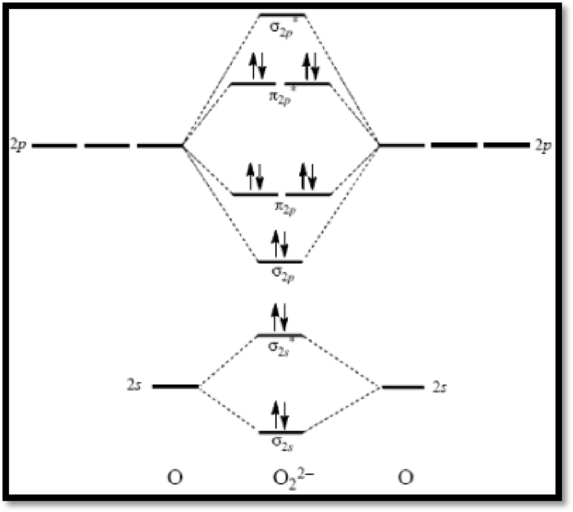
Figure 3
The bond order of
Peroxide ion
Want to see more full solutions like this?
Chapter 8 Solutions
Chemical Principles: The Quest for Insight
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





