
Interpretation:
A mechanism for the reaction of benzene with 2,2,5,5-teteamethyltetrahydrofuran shown below is to be proposed.
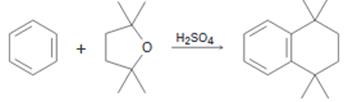
Concept introduction:
The reaction involves the electrophilc substitution of cyclic ether into a benzene ring. The electrophile can be produced by the protonation of the ethereal oxygen followed by a cleavage of a C-O bond. The eletrophilic attack will lead to the formation of an alcohol. Protonation of the alcohol and subsequent elimination of water generates another carbocation which can internally attack the ring. The loss of a proton from the intermediate will lead to product expected.
To propose:
A mechanism for the reaction of benzene with 2,2,5,5-teteamethyltetrahydrofuran shown.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- When 2-chloropropane treated with NaOH and 1-chloropropane treated with NaOH separately produce two different functional groups. Provide both reactions and explain the two different functional groups produced.arrow_forward4) Give an example of an enol which would tautomerize into a) an aldehyde and b) a ketone.arrow_forward10) For the reaction between isopropyl 1-propyl (or 'n-propyl') ether and HBr, which type of reaction best matches the expected products? A) Acid-catalyzed dehydration B) Nucleophilic substitution reaction C) Ether cleavage reaction D) Nucleophilic addition-elimination reactionarrow_forward
- Draw the overall reaction of 1-butanol with HBr and H2SO4 to form 1-bromobutane.arrow_forwardOutline syntheses of the following compounds, starting with triphenylphosphine, an alkyl halide and an aldehyde or ketone.a) 2-pentene b) Trans 1,2-diphenylethene c) Cyclohexylethenearrow_forwardSynthesize 1-methylcyclohexene from cyclohexanone and any organic alcohol.arrow_forward
- draw the products that could be formed in a reactionof benzene with 2hno3 in the presence of concentrated sulfuric acidarrow_forwardDescribe the procedures on how will you prepare an alcohol from an alkene.arrow_forwardWhat reaction conditions are needed to convert (R)-2-ethyl-2- methyloxirane to (R)-2-methylbutane-1,2-diol ?arrow_forward
- 1) What is the major product generally formed when a tertiary alkyl bromide (R3Br) reacts with sodium methoxide in methanol? 2) How is this major product formed?arrow_forward5) Compound A reacted with Mg in ether to give B. Compound B is with Ethyl bromide to give Ethyl cyclohexane. What are the structures of A and B?arrow_forwardProvide the structure of the major organic product of following ring opening reaction. CH;ONa CH;OHarrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
